 
Measuring the change in entropy for a system is relatively easy, but measuring the change in entropy for the surroundings is less easy to do directly. Using this formula we can judge if a reaction is spontaneous or not based on whether its change in entropy, and the surrounding accompanying change in entropy, facilitates a positive or negative change in entropy for the universe (following the second law). Together we get a formula for the overall change of entropy based on the system and surroundings: ΔSuniv = ΔSsyt + ΔSsur, where ΔSsyt is the change in the entropy of the system and ΔSsur is the change in the entropy of the surroundings. And the entropy of the surroundings (everything but the system itself) also being influenced by the entropy of the system. So we can imagine a reaction as a system whose entropy we care about. Usually when we do chemistry though we’re concerned not so much directly with the entropy of the universe, but rather the entropy change which accompanies a chemical reaction. This is why gas particles spread out in a container instead of concentrate themselves into a small area. The law and the formula essentially state that the universe naturally prefers greater levels of entropy and allows any process which causes a positive increase, but prohibits any process which causes a negative increase. A spontaneous process is one which happens naturally without the need of outside energy or work to help it along. So W is measuring how many energetically equivalent microstates a system can organize its energy into to get the same macrostate (or the system as we can observe it on a human scale).Īnother formula uses the second law of thermodynamics: ΔSuniv > 0, which put into words states that any spontaneous process increases the entropy of the universe (creates a positive change). A microstate is a particular way to order energy. Boltzmann’s formula uses a more mathematical approach to entropy by using W. The unit for entropy is the same as Boltzmann’s constant which means that entropy can also be understood as how many energy can be dispersed at a certain temperature since entropy is temperature dependent. 
One equation is Boltzmann’s equation: S = k*ln(W), where S is entropy (the usual variable for entropy), k is Boltzmann’s constant which is equal to the gas constant divided by Avogadro’s number which is approximately equal to 1.38 x 10^(-23) J/K, and W is the number of microstates which is a unitless quantity. There are several because entropy can be explained and used in a variety of ways. Since entropy is primarily dealing with energy, it’s intrinsically a thermodynamic property (there isn’t a non-thermodynamic entropy).Īs far as a formula for entropy, well there isn’t just one. The increase of disorder provides most of the free energy.First it’s helpful to properly define entropy, which is a measurement of how dispersed matter and energy are in a certain region at a particular temperature. ΔH The free energy comes mostly from a flow of thermal energy. ΔH > T * ΔS then the reaction is enthalpy-driven.The direction of a free energy change can be either enthalpy- or entropy-driven. ΔG0 - a nonspontaneous process - additional energy must put in for the reaction to happen (a round boulder being pushed up a hill).We can also define it with regards to the change in free energy: The Gibbs free energy equation is:Įarlier, we talked about spontaneity of a process and how it is associated with entropy. It's a function of both enthalpy and entropy, and is used to predict the spontaneity of a processes. What is Gibbs free energy? It's the energy in a system available to do work on its surroundings at constant pressure and temperature. 
Every system tends toward stability, and, for an irreversible process, maximum stability is reached it when the system's energy is most disordered. The entropy of a system is strictly connected to the systems energy. As stated by a physicist Rudolf Clausius: "The entropy of the universe tends to a maximum." You intuitively know that the opposite process is not possible - the milk won't separate from coffee by itself.Īny spontaneous process increases the disorder of the universe. You observed that the milk quickly mixes with the coffee. 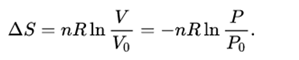
Let's say you've made yourself a hot cup of coffee. It might sound complicated, but you'll understand it easily with an everyday example. It doesn't have to be a fast - it can even be still occurring when the heat death of the universe occurs - but if it would proceed without the addition of any outside energy, it's spontaneous. It's one of the main determinants of the spontaneity of a reaction.Ī spontaneous process is one that doesn't require an outside source of energy to proceed. But why measure disorder, and is it even possible? Physically, we can't measure entropy, but we can calculate it. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
